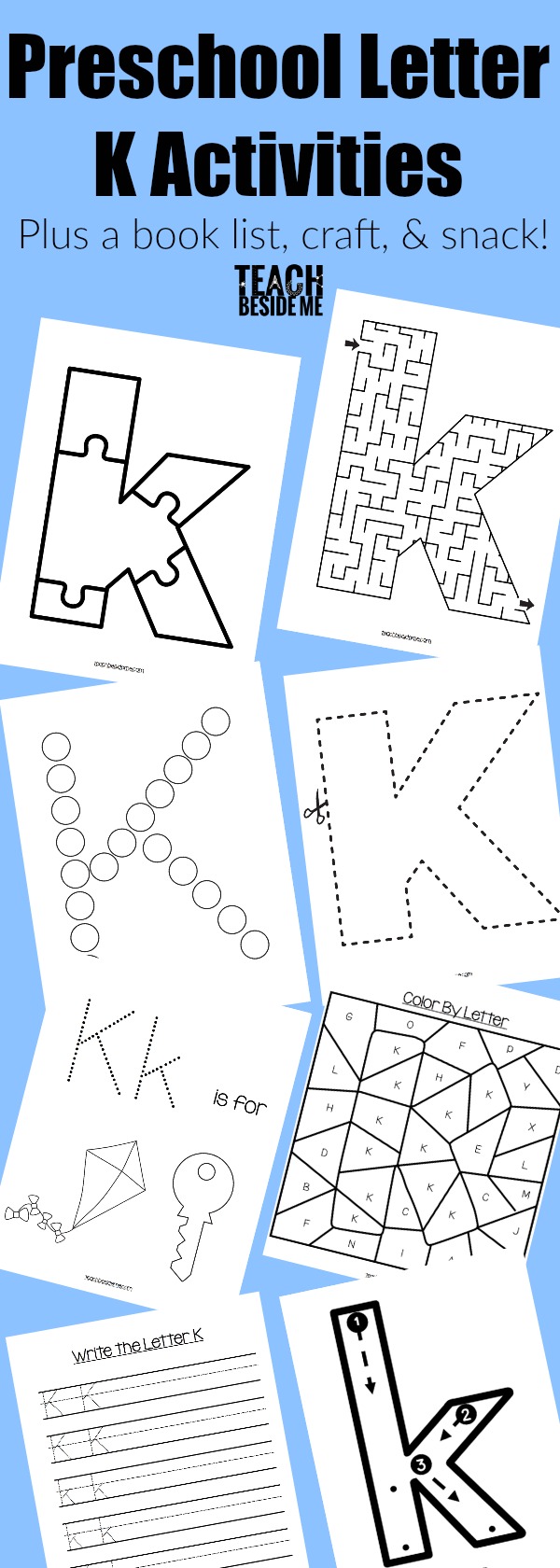
Harry Potter Dry Ice Crystal Ball Experiment
This post may contain affiliate links.
Have you been following along with the Harry Potter STEM series this week? Today’s project is a Harry Potter Crystal Ball Dry Ice Experiment! This experiment is also called a dry ice bubble.

These dry ice bubbles (crystal balls) could not be more exciting for kids! We played with this for over an hour and all of the kids stayed interested and involved the entire time!

Watch the Dry Ice Bubble in Action Here:
These kids had so much fun, as you can see! They wanted to get all dressed up in Harry-Potter themed costumes for the experiments.

How to Do the Dry Ice Experiment

You will need the following supplies to make the dry ice bubble:
Dry Ice- it is sold by the pound at grocery stores
Large bowl with a lip on the top
A long thing strip of fabric- knit or stretchy tends to work best
Soapy water (dish soap works just fine)
Warm water
Gloves to protect your hands
To make the Dry Ice Bubble- or the Harry Potter Crystal Ball- you need to fill the bowl about halfway full of warm water.
You should never touch dry ice. Make sure to always wear gloves when handling the dry ice. It can burn your skin.
Drop a chunk of dry ice into the water. When you drop the pieces of dry ice into the water it will create a ton of steam as it changes from a solid to a gas.

Fill a small cup with warm soapy water. Dip your fingers in the water and run them around the rim or lip of the bowl. Now dip the fabric into the soapy water squeezing out excess.

Slowly pull the fabric strip across the top of the bowl. You will see it catch and begin to make a bubble along the top of the bowl trapping in all of the steam created by the dry ice reaction.

Be very careful as you pull it off the end of the bowl, that’s where our bubble burst a lot of times!
Let the dry ice bubble grow and expand until it pops, or you pop it!

Repeat 2000 times.
If the steam begins to slow, add more dry ice into the water or more water to make sure it is covering the ice. After a little while, the water got soapy and we had to replace it with fresh water to keep the reaction going.

What is the Science Behind the Dry Ice Bubble Experiment?
Dry ice is frozen carbon dioxide. It is much colder than water, so it creates a bit of a reaction. When it is dropped into water, it is changing from a solid to a gas. Dry ice does not melt into a liquid form, only to a gas. This process is called sublimation. The vapor you see is both carbon dioxide and water vapor.
When you make a film of soap over the bowl, the gasses are trapped inside the bowl and accumulate until the bubble pops.
Check out even more Harry Potter STEM by popping over to Babble Dabble Do. She is hosting this fun series this week! Did you catch my Glowing Invisible Ink post?





You know, it’s a big problem to decide where to pin these, with the science topic it goes with, or on my Harry Potter board….
Hello! what kind of gloves do you need?
Something thick- I used some leather work gloves, or heavy winter gloves.